Past, present and future of lithium-ion batteries
M. Stanley Whittingham, recently awarded with the Nobel Prize in Chemistry, explains why lithium-ion batteries are so important for future mobility and what we can expect over the next few years.
Batteries account for 40% of the added value of electric vehicles and the expertise involved in their production plays an ever more decisive role within the Volkswagen Group. Their development is crucial - an increase in energy density, or energy storage capacity, and a reduction in costs are key to the widespread introduction of electric mobility.

M. Stanley Whittingham was awarded the Nobel Prize in Chemistry in 2019 for laying the scientific foundation for the lithium-ion batteries commonly used today. His research began in the early 1970s in the United States.
Whittingham attended this year's Science Award for Electrochemistry, which was set up by Volkswagen and BASF in 2012 to support scientific and engineering achievements in electrochemistry and provide impetus for the development of high-performance energy storage systems. During a trip to the Volkswagen Centre of Excellence in Salzgitter, the Nobel Laureate was able to see how his discovery is being put into practice in the pilot production of battery cells.
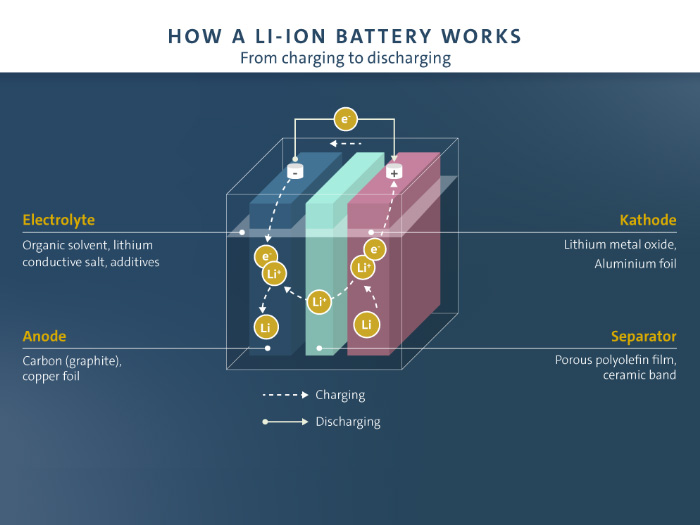
How does your scientific research relate to the battery cells that are built today?
I would say, I worked on the first experimental aspects. You know, can we make a lithium battery? How do we make it? And we did it on a very very small scale, starting back with single crystals in 1972. So, we’ve come from those little guys, to this huge effort. Transport is being transformed. So, it’s been a great journey from the idea to reality.
Are there any practical solutions that really surprised you?
Well I think it’s amazing, switching from the really challenging times – both in the battery area, and then for Volkswagen a few years ago – to seeing, really, electric vehicles happen, and seeing how we have modern methods in making these batteries, far beyond what we ever dreamed of.
How do you project the further development of lithium batteries? Are they going to be just better and better or is there a physical and chemical limit?
I think we can make the batteries at least double the energy density of those today. You know, we can’t go much beyond that. It’s not like Moore’s law for semi-conductors (note: which states that the speed of computer chips doubles every two years), so there is an ultimate limit. But we should be able to get to twice the energy density. So, twice the range, from the same size of battery that we have today. And hopefully, no more expensive. So we cut down the costs , increase the energy density, make them safer. We can all have one.
Today we use lithium ion batteries. What are the most interesting new approaches you would bet on?
I think it’s going to be lithium for the next ten, or twenty years. The question is: Can we get rid of the organic electrolyte? Than we can devise a battery that we can use pure lithium metal and maybe use a salt or electrolyte, be it inorganic or polymeric. So, I think that’s where the search is going to go, and that’s where most of us would put our money for twenty years out.
Are there any concepts that might sound like science fiction today, but from your point of view could soon be part of everyday life?
Well, I think, 50 years ago when we started out, with say these cars behind me, it would be science fiction. But now we’ve made it. So I think we’ve got to believe that we can do it. We must believe in the unbelievable. Then we can make it happen.
Source: Volkswagen AG
